Trials
KDSmart is designed for collecting data for trials. Trials can either be imported from external sources, transferred from KDXplore, or created within KDSmart.
Trials in KDSmart can be viewed and managed from the Trials screen:
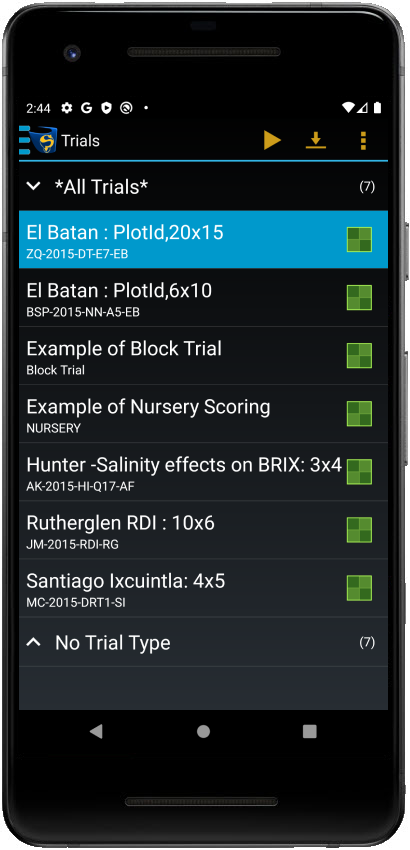
This page provides more information on trials, including creating and managing them in KDSmart.
What is a Trial?
Trial is the general term used in KDDart to refer to studies, experiments, nurseries, projects, etc. Trials contain data on the physical layout of a field and trial details and the plots and sub-plots within that field.
Trials also contain traits which are the qualities or characteristics being scored, and possibly tags that are reusable notes.
Generally, a trial is created in KDXplore or KDManage, transferred to KDSmart for scoring in the field, transferred back to KDXplore for data curation, and finally uploaded into KDDart.
Tip
Trials contain multiple plots - areas, spaces or even pots, uniquely identified by a plot ID, coordinate pairs (column/row ID), or both. Subplots are individual specimens within a plot.
Trial Types
There are three types of trials in KDXplore:
Column and Row (X/Y) - the combination of column and row numbers is a unique identifier for every plot in the trial;
Plot ID - the plot ID is a unique identifier for every plot; and
Block - the combination of block, column, and row numbers is a unique identifier for each plot.
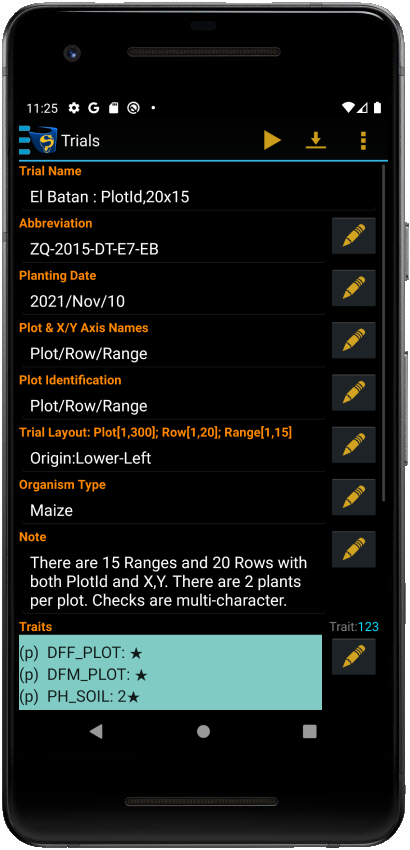
Trial Details
Each trial contains a set of attributes that define its characteristics. You can find the details for each trial in the Trial Details screen:
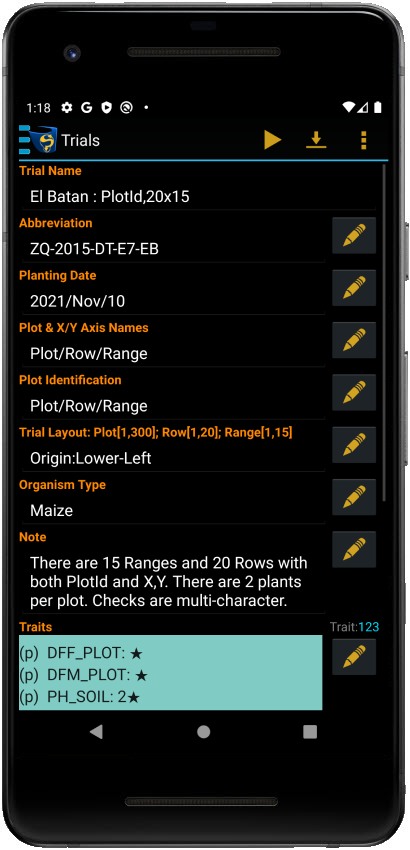
Trial Management
Trials in KDSmart are managed from the Trials screen, shown in the sections above. This screen has options for many trial management activities such as importing, exporting, creating, and editing trials.
Creating a New Trial
New X/Y and Plot ID trials can be created within KDSmart.
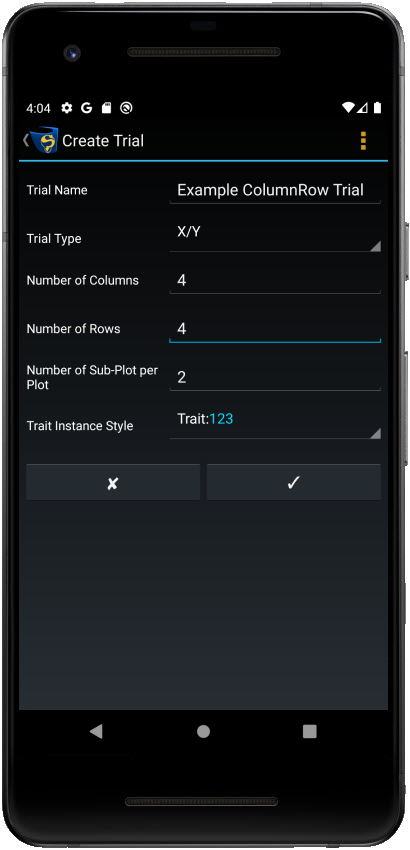
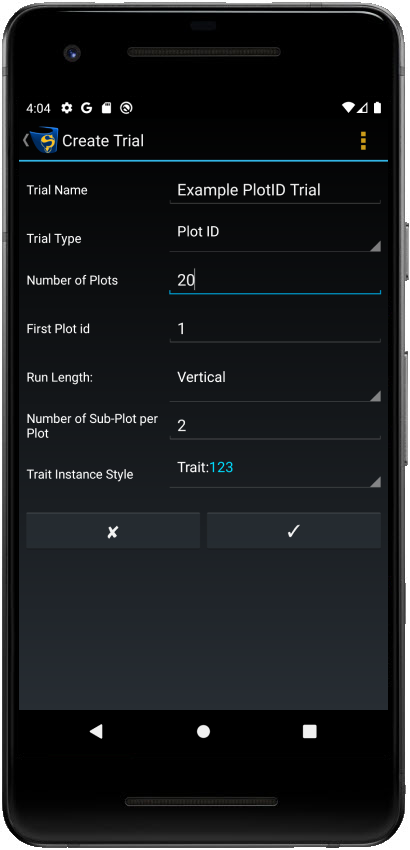
Step |
Action |
|---|---|
1. |
Navigate to the Trials screen and select the Menu button to open the Menu. |
2. |
Select the Create Trial button to display the Create Trial screen. |
3. |
Enter the details for the new trial. Depending on whether you are creating an X/Y or Plot ID trial, there will be different options. See the table below for a complete list of all details. |
4. |
Select the Confirm button to finalise the creation of the trial. |
Note
Currently, you cannot create new block trials in KDSmart. Instead, you will need to import or transfer them into the app.
Adding & Removing Traits in a Trial
Trials need to have traits added to them for scoring. You can also remove traits from an existing trial.
Step |
Action |
|---|---|
1. |
Select the Trial Details button to view the details of a trial. |
2. |
Select the Edit Traits button to display the Trait Instances screen. If there are existing traits, it will list them here. See the Preparing for Scoring page for information on this screen. |
3. |
Select the Menu button, and choose traits to add from under the title Trait Bundles. They are organised in bundles (if you have any bundles). This allows you to add sets of related traits at once. |
4. |
Select the Confirm button to add the traits. You will now see the newly added traits in the Trait Instances screen. |
To remove traits, go to the Trait Instances screen and select the Manage Traits button to open a menu where you will find the Remove option.
Note
If you don’t want to score a trait, it is not always necessary to completely remove it from the trial. See the Preparing for Scoring page for information on how to deselect traits when scoring.
Editing Trial Details
Trial details are displayed in the Trial Details screen. You can edit some details; however, trial attributes that are imported data are not editable. Each editable detail will have an Edit button next to it.
Deleting Trials
Long-press on a trial in the Trials screen to select it and then select the Delete option.